Our university has also decided anyone working in the summer doesn't require hall lights so this morning I wandered down a pitch black hall with one flickering fluorescent light. It's a wonder Jason, wolfman or bigfoot didn't pop out and spirit me away to some evil underground layer (as horror movie premises dictate).
So with all those lovely thoughts in mind I present today's topic of joy and happiness: blood.
And not just any blood, but sterile sheep blood. After a few false starts I was finally able to create my very own fancy pants blood plates with special supplements.
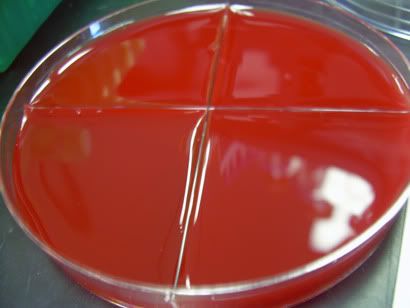 Plates are a giant pain in the ass. You measure out some really nasty smelling powder into a flask and then autoclave it to not only sterilize the media but to also get the damn thing into solution so you can pour it into the plates.
Plates are a giant pain in the ass. You measure out some really nasty smelling powder into a flask and then autoclave it to not only sterilize the media but to also get the damn thing into solution so you can pour it into the plates.Once the flasks come out of the autoclave they are a ticking time bomb (this is doubly so if you're working in small amounts) for as soon as it cools down it's going to solidify but instead of in the culture dishes it'll make a big gooey mess in the bottom of your flask.
This was my first time pouring blood plates so I kinda had a brain fart. While the flasks were staying warm in the water bath I set everything up and pulled the blood straight out of the refrigerator (this is the important part).
I collected everything together and then added 5mL's of blood (which made a huge CSI: crime scene out of the hood I might add). It looked pretty cool actually, the blood did a weird bloom thing:
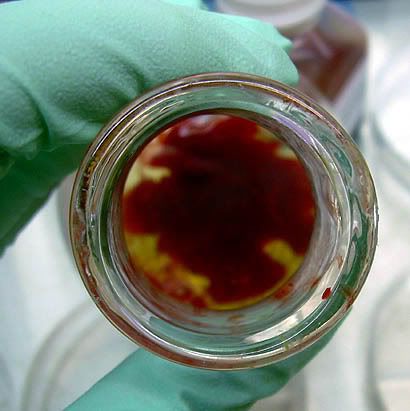
 You can already tell that something bad is happening around the edges. All that cold blood is diffusing, encouraging the nice warm agar to just give up its latent heat and lay down roots. In a matter of seconds I had a huge bloody mess coagulated in the flasks.
You can already tell that something bad is happening around the edges. All that cold blood is diffusing, encouraging the nice warm agar to just give up its latent heat and lay down roots. In a matter of seconds I had a huge bloody mess coagulated in the flasks.But after a long washing and cursing session at the sink I realized my mistake right away (I hate pouring 100 mL's worth of media. You've got less than 3 minutes really) and put the blood in the water bath to warm up while the blazing hot media cooled down.
Using warm blood, I was finally able to pour the plates and made another huge mess all over the hood. Blood is a real pain in the butt to clean up, I might add, and doubly so when it's trapped inside of agar.
Is it any wonder that just about every microbiologist you meet is a few bacterium short of a colony? It's a normal day when you have to mop up spilled blood and the trick is to make sure it's warm. I feel like I should be a member of the cast of Sweeny Todd sometimes.
Oh dear I think I see Freddy Krueger outside the window torturing one of the remaining vet students. I best go help.

3 comments:
Ugh, I have to pour regular plates today, and I am not looking forward to it (though we do large batches and pour at least 50 plates at a time, so it takes forever). I am going to try to push it off on an undergrad, muwahaha.
At my old job each month I had to pour over 13 L a day of TAP plates. That really sucked.
Are you doing the dreaded LB? I hate the smell of those. Though it's doubly worse when you autoclave the trash. YUCK!
You have the coolest job... its so NCIS/CSI forensic scientisty... I'm jealous!
RelentlessBride
Post a Comment